 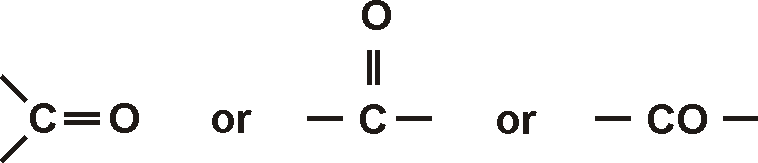
The solubility of carboxylic acids decreases with an increase in the size of the alkyl group or molecular masses because of reduced polarity and hindrance provided by large bulky groups to the carboxylic group for taking part in hydrogen bonding. The Solubility of lower members of carboxylic acids is due to hydrogen bonding. Benzoic Acid is soluble in cold water but it is soluble in hot water, ether. The first four members are very soluble in water and solubility decreases with rising molecular masses. The first 3 members of carboxylic acids are colorless liquids and have pungent smells. The physical properties of carboxylic acids are: This is why aldehydes and ketones have a higher boiling point than hydrocarbons. There are also attractive forces between permanent dipoles and molecules. The presence of carbon-oxygen double bonds in both aldehydes and ketones. 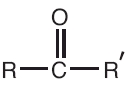
The boiling points of aldehydes and ketones increase as the number of carbon atoms increases. Boiling points of aldehydes and ketones are low than the alcohols and carboxylic acids of the molecular masses.Īmong these carbonyl compounds, Ketones have higher boiling points than isomeric aldehydes because of the presence of two electrons releasing groups around carbonyl carbon which makes them more polar.Īs the molecule lengthens, the number of electrons increases, and the attraction between the molecules increases. Therefore, Dipole-Dipole interactions are weaker than the intermolecular hydrogen bonding in alcohols and carboxylic acids. ISRO CS Syllabus for Scientist/Engineer Exam.ISRO CS Original Papers and Official Keys.GATE CS Original Papers and Official Keys.Full Stack Development with React & Node JS(Live).Java Programming - Beginner to Advanced.OS DBMS CN for SDE Interview Preparation.Data Structure & Algorithm-Self Paced(C++/JAVA).Full Stack Development with React & Node JS (Live).Data Structure & Algorithm Classes (Live). 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
